See on Scoop.it – PHYSICAL SCIENCES BREAK 1.0
The main assumptions of the kinetic theory of gases are as follows:
Gases are made up of particles (e.g. atoms or molecules). The size of these particles is very small compared to the distance between the particles.
These particles are constantly moving because they have kinetic energy. The particles move in straight lines at different speeds.
There are attractive forces between particles. These forces are very weak for gases.
The collisions between particles and the walls of the container do not change the kinetic energy of the system.
The temperature of a gas is a measure of the average kinetic energy of the particles.
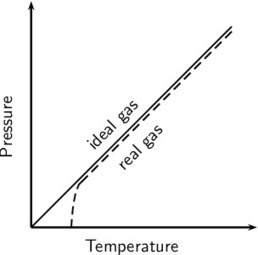
From these assumptions we can define the pressure and temperature of any gas.
Definition 1: PressureThe pressure of a gas is a measure of the number of collisions of the gas particles with each other and with the sides of the container that they are in.Definition 2: TemperatureThe temperature of a substance is a measure of the average kinetic energy of the particles. www.maimelatct.wordpress.com
See on everythingscience.co.za

Leave a Reply